How China and the West manage pharmaceuticals differently
Views: 15
From active ingredients to finished pills, the cost gap explained.
Frans Vandenbosch 方腾波 02/03/2026

Contentious, complicated, controlled
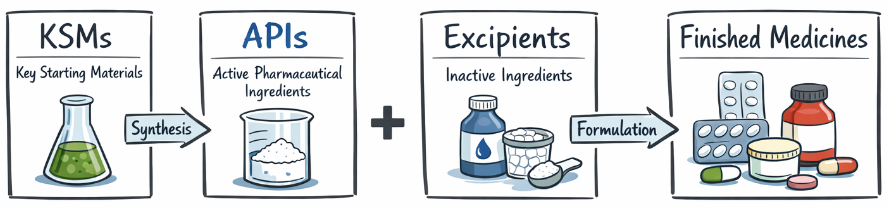
A medicine is a chain: feedstocks processed into key starting materials, synthesised into an active ingredient, combined with excipients, then manufactured, tested, packaged and shipped. Each link has a geography, a price and a regulatory condition attached to it.
Understanding why the same corticosteroid costs 46 times more in the United States than in China requires understanding the whole chain. The upstream chemistry is largely identical. Western companies buy their key starting materials and active ingredients from Chinese suppliers at prices comparable to what Chinese domestic firms pay. The divergence does not begin in the factory. It begins downstream, in the structures that govern how medicines are priced and reimbursed.
China has spent the past decade systematically preventing prices from drifting upward. The EU remains a patchwork of national systems negotiating separately, without shared buying power against some of the world’s largest corporations.
This article examines both sides of that contrast, starting with the science of what a medicine contains, then the gigantic scale of Chinese pharmaceutical production and finally how procurement policy, far more than production cost, explains what patients pay.
The global pharma industry
Before to dig in the global pharma industry, we need to understand how medicines are made:
Every medicine contains an Active Pharmaceutical Ingredient, or API. This is the actual drug substance, the molecule that produces the therapeutic effect. APIs are themselves built from key starting materials (KSMs), the chemical compounds assembled through synthesis to create the final active molecule. Think of KSMs as the raw building blocks, and the API as the finished structure they produce.
Yet an API alone is not a medicine. A pure active compound is typically a fine powder or crystalline substance with properties that make it impossible to manufacture reliably, dose accurately or administer safely. It may be chemically unstable, practically insoluble, bitter to the point of being unswallowable or simply too potent in raw form to handle consistently at industrial scale.
This is where excipients become essential. Excipients are pharmacologically inactive substances combined with the API to create a finished drug product. They do not treat the condition themselves, but without them the API cannot do its job. They are the delivery system: their role is to ensure the right quantity of drug reaches the right place in the body at the right time, in a form the patient can actually take.
Excipients are far from simple fillers. They are precisely engineered components, each selected for a specific function. A tablet may contain 6, 8 or more [11] distinct excipients, each contributing something the others cannot. Some hold the tablet together. Some ensure it dissolves at the correct rate. Some protect the API from heat, moisture or stomach acid. Some make the medicine acceptable to swallow. Remove any one of them and the product may fail, either in manufacturing, in stability or in the body itself.
Excipients must pass the same rigorous safety and quality standards as the API. They are tested, approved and tightly regulated. A medicine is only as reliable as the excipient system surrounding its active ingredient.
Key roles and categories of excipients
The following are the primary categories of excipients. Each fulfils a critical role in pharmaceutical formulations.
- Fillers and diluents convert tiny quantities of active ingredient into tablets or capsules of a practical, manufacturable size. Common examples include lactose, microcrystalline cellulose and calcium phosphate.
- Binders hold powder mixtures together, forming stable granules and maintaining tablet integrity during manufacturing. Typical examples include starch, hydroxypropyl methylcellulose and polyvinylpyrrolidone.
- Disintegrants cause tablets or capsules to break apart rapidly in digestive fluids, releasing the active ingredient for absorption. Examples include croscarmellose sodium and crospovidone.
- Lubricants prevent powders from sticking to manufacturing equipment and improve powder flow. Magnesium stearate and silica are widely used.
- Coatings protect sensitive compounds from moisture and light, mask unpleasant tastes and can control precisely where in the gut a drug dissolves.
- Preservatives prevent microbial contamination in liquids, creams and multi-dose containers. Parabens and benzyl alcohol are frequently employed.
- Solubilisers and surfactants help dissolve poorly soluble active ingredients, making intravenous medications possible and improving oral absorption. Examples include polysorbates and cyclodextrins.
- Flavours, colours and sweeteners improve palatability and help patients distinguish between products, reducing medication errors.
Why excipients are so important
The importance of excipients in modern medicine cannot be overstated. Without excipients, industrial pharmaceutical production would be impossible. Raw active ingredients have physical properties unsuitable for high-speed manufacturing equipment. Excipients transform them into consistent formulations, ensuring uniformity across millions of doses.
They also protect sensitive compounds from oxygen, light, heat and humidity. Medicines retain their potency throughout their shelf life. Patients receive the effect the prescriber intended.
Bioavailability depends entirely on excipient selection. The right choice determines whether an active ingredient reaches therapeutic concentrations in the body. The wrong choice means it passes through unused. A drug candidate can fail not because the molecule is wrong, but because its excipient system is.
The word “inactive” refers only to the absence of direct therapeutic action. It says nothing about importance. These ingredients undergo the same rigorous safety testing as the active compounds themselves.
All made in China:
KSMs, APIs, excipients and finished medicines
Many of our “western” medicines are based on TCM (Traditional Chinese Medicine). Willow bark contains salicin, which the body converts into salicylic acid. It is the basis of Bayer’s Aspirin. The pox vaccine was discovered in China. Artemisinin (a malaria drug) was invented in China in 1970. It is the basis of modern malaria treatment worldwide.
More recently, Roxadustat (an anaemia drug) was developed in China. Fruquintinib and many other of our modern ‘western’ cancer drugs were invented in China. Zanubrutinib (Bruton’s tyrosine kinase BTK inhibitor), Tislelizumab (Tevimbra, a PD-1 immune checkpoint inhibitor) and many more have their origin in China.
Only last month, a report published in Cell Press Journal reported a breakthrough in treatment of progressive multiple sclerosis with CAR-T cell therapy in Wuhan. [9]
Last year 2025, Chinese researchers have achieved a significant, world-first milestone in regenerative medicine by using stem-cell therapy to functionally cure both type 1 and type 2 diabetes. [10]
KSMs, API’s and excipients
China is by far the world’s largest producer of basic chemical feedstocks of pharmaceutical intermediates. For specific, critical Key Starting Materials (KSMs) like those for antibiotics (e.g., 6-APA for penicillins) and contraceptives, China’s global share is estimated to be over 90%.
China produces roughly 40 to 50 % of all the global APIs. China is dominant in the genericAPI market, supplying approximately 70 to 80 % of the global output.
In the Chinese pharma industry, about 74% of API’s are generic, 26% innovative.
There are more than 400 large pharmaceutical excipient manufacturers in China, producing about half of the global high value excipient volume. India is the number two, also producing certain excipients for medicines.
The cost of medicines in China and the EU
Corticosteroid drugs (medicines based on cortisone) are on average 46 times more expensive in the USA than in China, and more than 20 times more expensive in the EU. For common painkillers and anti-inflammatories such as Ibuprofen and Paracetamol, the disparity is even more striking.
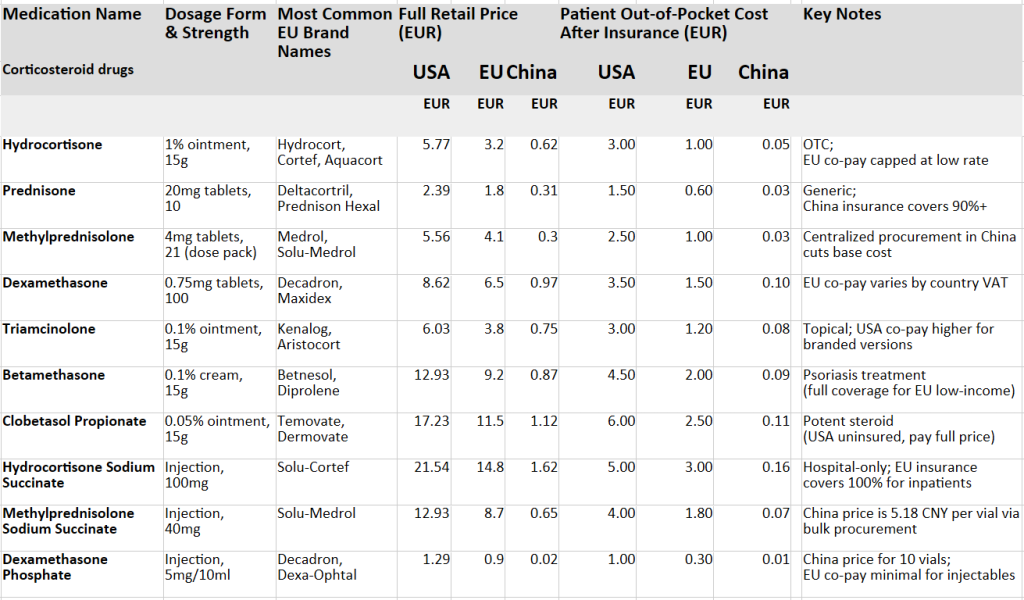

Chinese 醋酸地塞米松 (Dexamethasone acetate) and EU Dermovate
Western pharmaceutical companies source their active ingredients and key starting materials from the same Chinese suppliers at broadly comparable prices to domestic Chinese firms. The vast price gap in end markets is therefore almost entirely the product of mark-ups imposed by dominant Western corporations, a straightforward transfer of wealth from patients to shareholders.
China has systematically dismantled this logic within its own borders. Through its National Healthcare Security Administration, China conducts annual negotiations for the National Reimbursable Drug List, combined with centralised volume-based procurement that delivers guaranteed volumes in exchange for sharp price reductions. In 2024 alone, 38 globally-novel innovative drugs were negotiated into the national catalogue at a success rate exceeding 90%. The result: lower prices, stable supply, and no collapse in pharmaceutical innovation.
The European Union, by contrast, remains a patchwork of 27 separate negotiating tables. Each member state bargaining alone, without leverage, against the same well-resourced industry. Prices for identical medicines already vary tenfold between member states. The European Commission’s own assessment confirms that extended monopoly protections cost European health systems over €1.13 billion annually in entirely avoidable expenditure.
Coordinated European procurement modelled on China’s approach would achieve cost reductions not of 20 %, but of an entirely different order of magnitude. China has already proven this is operationally feasible at scale. European policymakers need only find the political will to negotiate together and to stop letting pharmaceutical corporations play member states off against one another.
Conclusion
Chinese and Western manufacturers buy the same active ingredients at the same prices. What happens next explains everything.
Western corporations apply mark-ups of 20 to 40 times. Not because of research costs, as many of these are decades-old generics. Not because of complexity, but because fragmented national health systems allow it. In the EU, identical medicines already vary tenfold in price between member states. That gap has nothing to do with science. It is pure extraction. The greed is difficult to ignore. When input costs are comparable but end prices differ by factors of twenty or more, the excess must go somewhere.
China chose not to accept this. Centralised negotiation, unified buying power and volume guarantees brought 38 new innovative drugs into the national catalogue in 2025, at a success rate exceeding 90 per cent. Access without plunder.
Europe could do the same. The mechanism exists. The proof exists. All that is missing is the political will to stop protecting an industry that has grown wealthy on the suffering of patients and taxpayers.
Thank you for reading! We’d love to hear your thoughts. Please share your comments here below and join the conversation with our community!
本文中文:
Dit artikel in het Nederlands: Hoe China en het Westen op verschillende manieren omgaan met geneesmiddelen
Endnotes
[1] Gao, B. (2024). Study on the Impact of Volume-Based Procurement Policy on the Productivity of Listed Pharmaceutical Companies in China. *Modern Management*.
[2] National Healthcare Security Administration. (2024, November 28). China’s drug price negotiation program saves billions for patients. *English.gov.cn*.
[3] Association of European Cancer Leagues & European Fair Pricing Network. (2024, September 11). Better access to medicines through reduced regulatory data protection and modulated incentives.
[4] European Parliament. (2023). Motion for a resolution on EU-wide joint procurement of medicines to reduce undue price differences between member states (B9-0006/2023).
[5] Yin, N. (2024, November 1). China’s Centralised Volume-Based Drug Procurement: Effectiveness and Lessons [Academic seminar]. Central University of Finance and Economics, Beijing, China.
[9] Anti-BCMA CAR-T therapy in patients with progressive multiple sclerosis https://www.cell.com/cell/fulltext/S0092-8674(25)01088-8
[10] Min xu & Guang Ning. Breakthrough in human induced pluripotent stem cell research of a functional cure for type 1 diabetes 06/01/2025
[11] Amoxicillin, a common antibiotic, contains 8 excipients: microcrystalline cellulose, sodium starch glycolate, colloidal silicon dioxide, magnesium stearate, talc, gelatin, titanium dioxide, sodium lauryl sulfate.



This is a very patient and clear explanation of the differences in the price and process of pharmaceuticals in China, Europe and the USA. Astounding. Many thanks.